s-Block ElementsHard
Question
Which of the following statement (s) is/are correct with respect to alkali metal bicarbonates ?
Options
A.In crystalline sodium bicarbonate, the HCO3- anions form an infinite chain through H-bonding.
B.Bicarbonates do not produce any colour with phenophthalein indicator.
C.All bicarbonates except that of lithium are found in solid state.
D.KHCO3 is formed when CO2 (in excess) reacts with KO2 (medium water).
Solution
(A) 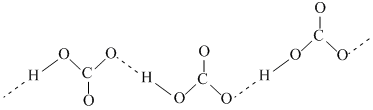
(B) As bicarbonate is a acidic salt.
(C) In solid, smaller Li+ polarises bigger CO32- and gets decomposed liberating CO2 gas. But LiHCO3 exists in aqueous solution.
(D) 4 KO2 + 4 CO2 (excess) + 2 H2O → 4KHCO3 + O2.

(B) As bicarbonate is a acidic salt.
(C) In solid, smaller Li+ polarises bigger CO32- and gets decomposed liberating CO2 gas. But LiHCO3 exists in aqueous solution.
(D) 4 KO2 + 4 CO2 (excess) + 2 H2O → 4KHCO3 + O2.
Create a free account to view solution
View Solution Free