p-Block elementsHard
Question
Iron exhibits +2 and +3 oxidation states. Which of the following statements about iron is incorrect ?
Options
A.Ferrous compounds are more easily hydrolysed than the corresponding ferric compounds.
B.Ferrous oxide is more basic in nature than the ferric oxide.
C.Ferrous compounds are relatively more ionic than the corresponding ferric compounds.
D.Ferrous compounds are less volatile than the corresponding ferric compounds.
Solution
Ferrous salt → Fe+2
Ferric salt → Fe+3
φ (Ionic potential)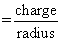
Fe+2 has low value of f compared to the Fe+3 , therefore
(1) Ferrous compounds will be less hydrolysed in comparison of Ferric.
(2) Ferrous oxide will be more basic than Ferric oxide.
(3) Ferrous compounds are relatively more ionic than Ferric compounds.
(4) Ferrous compounds have higher ionic character i.e. less volatile.
Ferric salt → Fe+3
φ (Ionic potential)
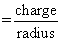
Fe+2 has low value of f compared to the Fe+3 , therefore
(1) Ferrous compounds will be less hydrolysed in comparison of Ferric.
(2) Ferrous oxide will be more basic than Ferric oxide.
(3) Ferrous compounds are relatively more ionic than Ferric compounds.
(4) Ferrous compounds have higher ionic character i.e. less volatile.
Create a free account to view solution
View Solution FreeMore p-Block elements Questions
Chlorine acts as a bleaching agent only in presence of :...The product obtained in the reaction of diborane with excess of ammonia at low temperature is :...The reaction of white phosphorous with aqueous NaOH gives phosphine along with another phosphorouscontaining compound. T...Sodium nitrate decomposes above 800oC to give :...Which of the following is only acidic in nature?...