p-Block elementsHard
Question
The reaction of white phosphorous with aqueous NaOH gives phosphine along with another phosphorouscontaining compound. The reaction type; the oxidation states of phosphorus in phosphine and the otherproduct are respectively
Options
A.redox reaction; -3 and -5
B.redox reaction; +3 and +5
C.disproportionation reaction; -3 and +5
D.disproportionation reaction; -3 and +3
Solution
The balanced disproportionation reaction involving white phosphorus with aq. NaOH is
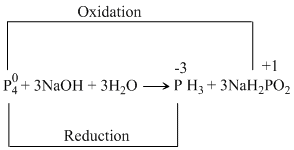
* However, as the option involving +1 oxidation state is completely missing, one might consider that NaH2PO2 formed has undergone thermal decomposition as shown below:

Although heating is nowhere mentioned in the question, the ″other product″ as per available options seemsto be Na2HPO4 (oxidation state = +5).

* However, as the option involving +1 oxidation state is completely missing, one might consider that NaH2PO2 formed has undergone thermal decomposition as shown below:

Although heating is nowhere mentioned in the question, the ″other product″ as per available options seemsto be Na2HPO4 (oxidation state = +5).
Create a free account to view solution
View Solution Free