p-Block elementsHard
Question
Which of the following statement(s) is / are correct ?
Options
A.Nitrogen (IV) oxide in basic aqueous solution disproportionates.
B.Sodium nitrate and lead nitrate each on heating to 800oC decomposes liberating both NO2 and O2 gases.
C.Ammonia and sodium hypochlorite react in dilute aqueous solutions in presence of gelatin and liberate nitrogen gas.
D.Hydrogen peroxide is produced on large scale by the auto oxidation of 2-ethyl anthraquinol.
Solution
(A) In basic solution NO2 disproportionates into N(III) and N(V), forming NO2- and NO3- ions.
2NO2 (aq) + 2OH- (aq) → NO2- (aq) + NO3- (aq) + H2O(l).
(B) 4NaNO3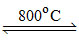 2Na2O + 5O2 + 2N2
2Na2O + 5O2 + 2N2
2Pb(NO3)2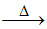 2PbO + 4NO2 + O2
2PbO + 4NO2 + O2
(C) NH3(aq) + NaOCl (aq)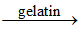 NH2Cl2 (aq) + NaOH (aq)
NH2Cl2 (aq) + NaOH (aq)
(dilute solutions)
2NH3(aq) + NH2Cl2 → N2H4 (aq) + NH4Cl
(D)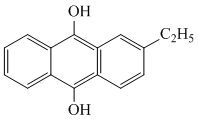
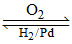
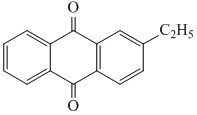 + H2O2
+ H2O2
2NO2 (aq) + 2OH- (aq) → NO2- (aq) + NO3- (aq) + H2O(l).
(B) 4NaNO3
2Pb(NO3)2
(C) NH3(aq) + NaOCl (aq)
(dilute solutions)
2NH3(aq) + NH2Cl2 → N2H4 (aq) + NH4Cl
(D)

 + H2O2
+ H2O2Create a free account to view solution
View Solution FreeMore p-Block elements Questions
Carbongene has X% of CO2 and is used as an antidote for poisoning of Y . Then, X and Y are...H3BO3 $\overset{\quad T_{1}\quad}{\rightarrow}$ X $\overset{\quad T_{2}\quad}{\rightarrow}$ Y $\overset{\quad redhot\qua...The following compounds have been arranged in order of their increasing thermal stabilities. Identify the correct order ...The chemical formula of ″tear gas″ is...Regarding the hydrides of group 15 elements ${EH}_{3}$ ( $E = N,P,As,Sb$ ), select the correct statement from the follow...