p-Block elementsHard
Question
Which of the following statement(s) is/are incorrect ?
Options
A.SbH3 > NH3 > ASH3 > PH3 (boiling point)
B.H3PO4 > H3PO3 > H3PO2 (reducing character)
C.N2O < NO < N2O3 < N2O5 (oxidation state on nitrogen atom)
D.NH3 > PH3 >ASH3 > SbH3 ≥ BiH3 (basicity)
Solution
Correct order is
H3PO2 > H3PO3 > H3PO4
as number of reducing hydrogen decreases. Redusing nature decreases.
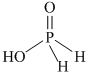
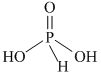
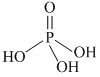
Note : Hydrogen atom directly attached to phosphorus is called reducing hydrogen.
H3PO2 > H3PO3 > H3PO4
as number of reducing hydrogen decreases. Redusing nature decreases.



Note : Hydrogen atom directly attached to phosphorus is called reducing hydrogen.
Create a free account to view solution
View Solution FreeMore p-Block elements Questions
When chlorine gas is passed through an aqueous solution of a potassium halide in the presence of chloroform, a voilet co...Thermodynamically, the most stable form of carbon is...Which of the following species is basic and reducing ?...The energy gaps (Eg) between valence band and conduction band, for diamond, silicon and germanium are in the order....Identify the correct order of solubility in aqueous medium :...