p-Block elementsHard
Question
Which of the following species is basic and reducing ?
Options
A.SO32-
B.SO42-
C.S2O42-
D.HSO4-
Solution
SO32- : 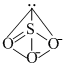 It contains one lone pair of electron in sp3 hybrid orbital. SO32- + 2H+ → SO2 + H2O.
It contains one lone pair of electron in sp3 hybrid orbital. SO32- + 2H+ → SO2 + H2O.
S2O42- :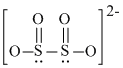
 It contains one lone pair of electron in sp3 hybrid orbital. SO32- + 2H+ → SO2 + H2O.
It contains one lone pair of electron in sp3 hybrid orbital. SO32- + 2H+ → SO2 + H2O.S2O42- :
Create a free account to view solution
View Solution FreeMore p-Block elements Questions
In the reaction O3 + 2I- + H2O → 2OH- + (X) + O2 . The compound (X) is :...Select the incorrect statement about the boron....Three allotropes (A), (B) and (C) of phosphorous in the following change are respectively...A tetra-atomic molecule (A) on reaction with nitrogen(I)oxide, produces two substances (B) and (C). (B) is a dehydrating...Extra pure N2 can be obtained by heating...