p-Block elementsHard
Question
The high reactivity and low volatility of white phosphorous is due to :
Options
A.tetrahedrally arranged P4 units
B.bond angle of 600 increases steric (strain) factor
C.weak van der Waals forces of attraction
D.both (B) and (C)
Solution
Has angle strain 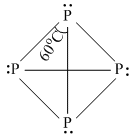 → sp3 (normal tetrahedral bond angle is 109.5o)
→ sp3 (normal tetrahedral bond angle is 109.5o)
 → sp3 (normal tetrahedral bond angle is 109.5o)
→ sp3 (normal tetrahedral bond angle is 109.5o)Create a free account to view solution
View Solution FreeMore p-Block elements Questions
KClO3 + H2SO4 KHSO4 + HClO4 + (X) + H2OIn above unbalanced chemical equation, the product (X) may be :...Which of the following product is formed by the reaction of sulphur dioxide with chlorine in presence of charcoal ?...An inorganic compound (A) made of two most occuring elements into the earth crust, having a polymeric tetra-headral netw...H2O is oxidised to O2 by :...The gas obtained on heating lead nitrate to 400oC is :...