p-Block elementsHard
Question
Silicone resins are made by :
Options
A.dissolving a mixture of PhSiCl3 and (Ph)2SiCl2 in toluene and then hydrolysis with water.
B.hydrolysing a mixture of (CH3)2SiCl2 and (CH3)3SiCl.
C.hydrolysis of (CH3)2SiCl2
D.none
Solution
(A) When a mixture of PhSiCl3 and (Ph)2SiCl2 is dissolved in toluene and then hydrolysed with water silicon resins are obtained
(B) Silicones from the hydrolysis of a mixture of (CH3)3 SiCl & (CH3)2 SiCl2
The dichloro derivative will form a long chain polymer as usual. But the growth of this polymer can be blocked at any stage by the hydrolysis product of mono-chloro derivative.
(C)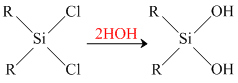 + 2HCl
+ 2HCl
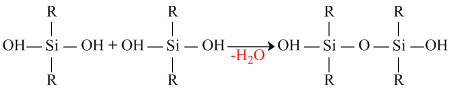
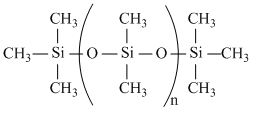
In this manner several molecules may combine to form a long chain polymer whose both the ends will be occupied by -OH groups. Such compounds are generally represented from the following formula.
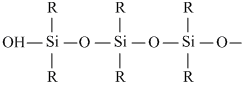
(B) Silicones from the hydrolysis of a mixture of (CH3)3 SiCl & (CH3)2 SiCl2
The dichloro derivative will form a long chain polymer as usual. But the growth of this polymer can be blocked at any stage by the hydrolysis product of mono-chloro derivative.

(C)
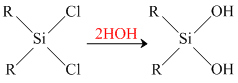 + 2HCl
+ 2HCl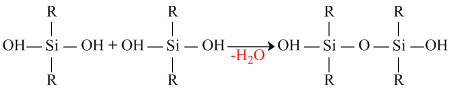
In this manner several molecules may combine to form a long chain polymer whose both the ends will be occupied by -OH groups. Such compounds are generally represented from the following formula.
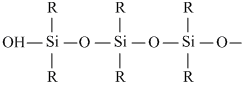
Create a free account to view solution
View Solution FreeMore p-Block elements Questions
Which of the following statements is/are correct regarding inter-halogen compounds of ABx types?...The mixture of concentrated HCl and HNO3 made in 3 : 1radio contains :...Metaphosphoric acid exists in polymeric form and may have :...Which of the following gives H2O2 on hydrolysis ?...H2S gas is not evolved when SO32- ion reacts with following reagents....