p-Block elementsHard
Question
Which of the following facts regarding boron and silicon is true ?
Options
A.Boron is used to make boron steel or boron carbide control rods for nuclear reactor.
B.Boron and silicon form halides which are not hydrolysed.
C.Boron and silicon react with magnesium to form magnesium boride and magnesium silicide which are decomposed by acids to give volatile borane and silane, respectively.
D.Both boron and silicon react with alkali to form borates and silicates containing BO33- and SiO44- tetrahedral units, respectively.
Solution
BCl3 + 3H2O 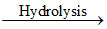 H3BO3 + 3HCl ; SiCl4 + 3H2O
H3BO3 + 3HCl ; SiCl4 + 3H2O 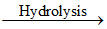 H2SiO3 + 4HCl
H2SiO3 + 4HCl
Create a free account to view solution
View Solution Free