Ionic EquilibriumHard
Question
A weak acid (HA) after treatment with 12 mL of 0.1 M strong base (BOH) has a pH of 5. At the end point, the volume of same base required is 27 mL. Ka of acid is (log2 = 0.3)
Options
A.1.8 × 10-5
B.8 × 10-6
C.1.8 × 10-6
D.8 × 10-5
Solution
m. moles of HA taken = 27 × 0.1 = 2.7
HA + OH- A- + H2O
t=0 2.7 1.2
teq 1.5 - 1.2
pH = pKa + log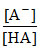 ⇒ 5 = pKa + log
⇒ 5 = pKa + log 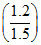 = pKa + log
= pKa + log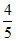
∴ pKa = 5.1 ⇒ Ka = 8 × 10-6.
HA + OH- A- + H2O
t=0 2.7 1.2
teq 1.5 - 1.2
pH = pKa + log
∴ pKa = 5.1 ⇒ Ka = 8 × 10-6.
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
A solution of Na2CO3 is added drop by drop to litre of a solution containing 10-4 mole of Ba2+ and 10-5 mole of Ag+, if ...An element X occurs in short period having configuration ns2np3. The formula and nature of its oxide is :-...HCN is weak acid (Ka = 4.0 × 10-4). Which statements is not true for an aqueous solution of hydrocyanic acid ?...At 25oC, the solubility product of Mg(OH)2 is 1.0 × 10-11. At what pH, will Mg2+ ion start precipitating in the for...Concentration of the Ag+ ions in a saturated solution of Ag2C2O4 is 2.2 ×10-4 mol L-1 Solubility product of Ag2C2O4...