Ionic EquilibriumHard
Question
Approximate pH of 0.1 M aqueous H2S solution when K1 and K2 for H2S at 25oC are 1 × 10-7 and 1.3 × 10-13 respectively :
Options
A.4
B.5
C.6
D.8
Solution
pH of 0.1 M H2S solution can be derived by :
H2S ⇋ H+ + HS- ; K1 = 1 × 10-7
∴ [H+] = Cα = C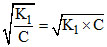
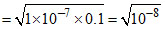 = 10-4
= 10-4
∴ pH = 4
H2S ⇋ H+ + HS- ; K1 = 1 × 10-7
∴ [H+] = Cα = C
∴ pH = 4
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
Calcium lactate is a salt of weak acid and represented as Ca(Lac)2. A saturated solution of Ca(Lac)2 contains 0.125 mole...The vapour pressure of a given liquid will decrease if....Which of the following are true for an acid-base titration ?...Which of the following relation(s) is/are true for alkaline solution?...What fraction of an indicator HIn is in basic form at a pH of 6 if the pKa of the indicator is 5 ?...