Periodic TableHard
Question
The correct order of the decreasing radii among the following isoelectronic species is
Options
A.Ca2+ > K+ > S2- > Cl-
B.Cl- > S2- > Ca2+ > K+
C.S2- > Cl- > K+ > Ca2+
D.K+ > Ca2+ > Cl- > S2-
Solution
Ionic radii ∝ charge on anion ∝ 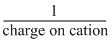
During the formation of a cation, the electrons are lost from the outer shell and the remaining electrons experience a great force of attraction by the nucleus, ie, attracted more towards the nucleus. In other words, nucleus hold the remaining electrons more tightly and this results in decreased radii.
However, in case of anion formation, the addition of electron(s) takes place in the same outer shell, thus the hold of nucleus on the electrons of outer shell decreases and this results in increased ionic radii. Thus, the correct order of ionic radii is
S2- > Cl- > K+ > Ca2+
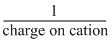
During the formation of a cation, the electrons are lost from the outer shell and the remaining electrons experience a great force of attraction by the nucleus, ie, attracted more towards the nucleus. In other words, nucleus hold the remaining electrons more tightly and this results in decreased radii.
However, in case of anion formation, the addition of electron(s) takes place in the same outer shell, thus the hold of nucleus on the electrons of outer shell decreases and this results in increased ionic radii. Thus, the correct order of ionic radii is
S2- > Cl- > K+ > Ca2+
Create a free account to view solution
View Solution FreeMore Periodic Table Questions
Ionic radii of :...Beryllium and aluminium exhibit many properties which are similar. But the two elements differ in...Which of the following is the correct statement :(a) Boron is diagonally related to silicion(b) Elements of third group ...Given below are two statements :Statement-I : $C < O < N < F$ is the correct order in terms of first ionization...In period 4 of the periodic table, the elements with highest and lowest atomic radii are respectively....