Periodic TableHard
Question
Ionic radii of :
Options
A.Ti4+ < Mn7+
B.35Cl-, 37Cl-
C.K+ > C-
D.P3+ > P5+
Solution
(i) Higher is the charge on cation, smaller is the size so P3+ > P5+ and not Ti4+ < Mn7+
(ii) Cation is always smaller than isoelectronic anion so K+ > Cl-
(iii) Number of neutron do not affect radii so35Cl-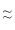 37Cl-
37Cl-
(ii) Cation is always smaller than isoelectronic anion so K+ > Cl-
(iii) Number of neutron do not affect radii so35Cl-
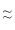 37Cl-
37Cl-Create a free account to view solution
View Solution Free