Ionic EquilibriumHard
Question
The pH of a 0.1 molar solution of the acid HQ is 3. The value of the ionization constant, Ka of this acid is :-
Options
A.1 × 10 -7
B.3 × 10 -7
C.1 × 10 -3
D.1 × 10 -5
Solution
pH = -log[H+] pH = 3
[H+] = 10-pH = 10-3
For weak acid
[H+] =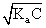
[H+]2 = Ka × C
(10-3)+ = Ka × 0.1
Ka = 10-5
[H+] = 10-pH = 10-3
For weak acid
[H+] =
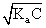
[H+]2 = Ka × C
(10-3)+ = Ka × 0.1
Ka = 10-5
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
Which of the following mixtures will act as buffer ?...The solubility of sparingly soluble salt A3B2 (molar mass = ‘M’ g/mol) in water is ‘x’ g/L. The ratio of molar concentra...Consider a titration of potassium dichromate solution with acidified Mohr′s salt solution using diphenylamine as i...The molar solubility of Zn(OH)2 in 1 M ammonia solution at room temperature is (Ksp of Zn(OH)2 = 1.6 × 10−17; Kstab of Z...The strength of an aqueous solution of I2 can be determined by titrating the solution with standard solution of:...