Ionic EquilibriumHard
Question
Which of the following species is more soluble in water:
Options
A.M(OH)3 (KSP = 1 × 10-35)
B.M(OH)2 (KSP = 1 × 10-30)
C.MOH (KSP = 1 × 10-28)
D.MOH (KSP = 1 × 10-26)
Solution
solubility of M(OH)3 = 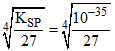 = 7.8 × 10-10
= 7.8 × 10-10
solubility of M (OH)2 =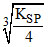 = 6.3 × 10-11
= 6.3 × 10-11
solubility of MOH =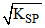 = 10-14
= 10-14
solubility of M (OH)2 =
solubility of MOH =
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
The pH of water is 7.0 at 25oC. If water is heated to 70oC, then...The equilibrium constant for the ionization of RNH2(g) in water as RNH2(g) + H2O(l) $\rightleftharpoons$ RNH3+ (aq) + O...Which of the following solutions when added to 1L of a 0.01 M CH3COOH solution will cause no change in the degree of dis...If the pH of 0.001 M potassium propionate solution be 8.0, then the dissociation constant of propionic acid will be...A solution contains a mixture of Ag+ (0.10 M) and Hg22+ (0.10 M), which are to be separated by selective precipitation. ...