Ionic EquilibriumHard
Question
Slaked lime, Ca(OH)2 is used extensively in sewage treatment. What is the maximum pH that can be established in Ca(OH)2 (aq):
Ca(OH)2 (s) ⇋ Ca2+ (aq) + 2OH- (aq) ; KSP = 5.5 × 10-6
Ca(OH)2 (s) ⇋ Ca2+ (aq) + 2OH- (aq) ; KSP = 5.5 × 10-6
Options
A.1.66
B.12.35
C.7.0
D.14.0
Solution
KSP of Ca(OH)2 = 4s3 = 5.5 × 10-6
∴ s = 3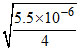 = 1.11 × 10-2
= 1.11 × 10-2
∴ [OH-] = 2s = 2 × 1.11 × 10-2
∴ pOH = 1.65 ∴ pH = 12.35
∴ s = 3
∴ [OH-] = 2s = 2 × 1.11 × 10-2
∴ pOH = 1.65 ∴ pH = 12.35
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
Most reactive towards Nucleophilic attack -...Incorrect order of ionic character :...The vapour pressure of a given liquid will decrease if....2-Hexyne gives trans -2-Hexene on treatment with :-...Two buffer solutions, A and B, each made with acetic acid and sodium acetate differ in their pH by one unit, A has salt ...