Chemical BondingHard
Question
The correct order of increasing bond angles in the following species is
Options
A.Cl2O < ClO2 < ClO2-
B.ClO2 < Cl2O < ClO2-
C.Cl2O < ClO2- < ClO2
D. ClO2- < Cl2O < ClO2
Solution
As the number of ione pair of electrons increases, band angle decreases due to repulsion between lp - lp. Moreover, as the electro negativity of central atom decreases, bond angle decreases.
Hence, the order of bond angle is
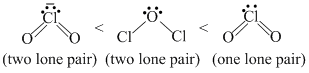
(Cl is less electronegative as compared to O.)
Hence, the order of bond angle is
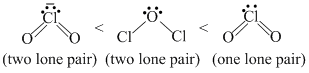
(Cl is less electronegative as compared to O.)
Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
Bond Angle is maximum in :-...Element M forms which types of sulphate :-...Which pair have identical bond order :-...If a molecule MX3 has a zero dipole moment, the sigma bonding orbitals used by M (atomic number < 18) :-...In allene (C3H4), the type(s) of hybridization of the carbon atoms is (are)...