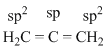Chemical BondingHard
Question
In allene (C3H4), the type(s) of hybridization of the carbon atoms is (are)
Options
A.sp and sp3
B.sp and sp2
C.only sp2
D.sp2 and sp3
More Chemical Bonding Questions
Identify the molecule $(X)$ with maximum number of lone pairs of electrons (obtained using Lewis’s dot structure) among ...Addition of phosophate fertilisers to water bodies causes :...Which of the following is para-magnetic ?...The ion that is isorlrctronic with CO is :...Which has Largest value of dipole moment is :-...