Ionic EquilibriumHard
Question
Which of the following correctly explains the nature of boric acid in aqueous medium:
Options
A.H3BO3 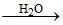 H3+O + H2BO3-
H3+O + H2BO3-
B.H3BO3 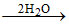 2H3+O + HBO32-
2H3+O + HBO32-
C.H3BO3 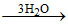 3H3+O + BO33-
3H3+O + BO33-
D.H3BO3 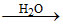 B(OH)4- + H+
B(OH)4- + H+
Solution
H3BO3 is weak, Lewis monobasic acid and shows the given equilibrium.
H3BO3 + H2O ⇋ B(OH)4- + H+
H3BO3 + H2O ⇋ B(OH)4- + H+
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
pH of 0.01 M-(NH4)2SO4 and 0.02 M-NH4OH buffer (pKa of NH4+ = 9.26) is...Number of H+ ions present in 10 mL of solution of pH = 3 are:...What is the solubility of MnS in pure water, assuming hydrolysis of S2– ions?Ksp of MnS = 2.5 × 10–10, Ka1 = 1 × 10–7 an...For pure water...What concentration of free CN– must be maintained in a solution that is 1.8 M-AgNO3 and 0.16 M-NaCl to prevent AgCl from...