Ionic EquilibriumHard
Question
A solution is a mixture of 0.05 M KCI and 0.05 M NaI. The concentration of I- in the solution when AgCI just starts to precipitate is equal to: (KSPAgCI = 10-10 M2 ; KSPAgI = 4 × 10-16 M2)
Options
A.4 × 10-6 M
B.2 × 10-8 M
C.2 × 10-7 M
D.8 × 10-15 M
Solution
For AgCI precipitation [Ag+] = 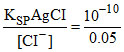 = 2 × 10-9
= 2 × 10-9
For AgI precipitation [Ag+] =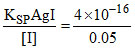 = 8 × 10-15
= 8 × 10-15
Thus AgI will precipitate first. AgCI will precipitate only when,
[Ag+] = 2 × 10-9; Thus [I-]Left =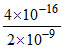 = 2 × 10-7 M
= 2 × 10-7 M
For AgI precipitation [Ag+] =
Thus AgI will precipitate first. AgCI will precipitate only when,
[Ag+] = 2 × 10-9; Thus [I-]Left =
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
Which of the following statements about a weak acid strong base titration is/are correct ?...If Ka1 and Ka2 of H2SO4 are 10-2 and 10-6 respectively then...The relation pKa = 14 – pKb is true for which of the following pair(s)?...Which is most stable ?...A saturated solution of silver benzoate (AgOCOC6H5) has pH of 8.6. Ka for benzoic acid is 5.0 × 10–5. The value of Ksp f...