Gaseous StateHard
Question
The standard reduction potential data at 25oC is given below
Eo (Fe3+, Fe2+) = + 0.77 V ;
Eo (Fe2+, Fe) = - 0.44 V ;
Eo (Cu2+, Cu) = + 0.34 V ;
Eo (Cu+, Cu) = + 0.52 V ;
Eo (O2(g) + 4H+ + 4e- → 2H2O] = +1.23 V ;
Eo [(O2(g) + 2H2O + 4e- → 4OH- )] = +0.40 V ;
Eo (Cr3+, Cr) = -0.74 V ;
Eo (Cr2+, Cr) = -0.91 V ;
Match Eo of the redox pair in List-I with the values given in List-II and select the correct answer using the code given below the lists :
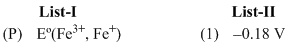
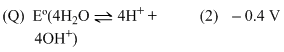
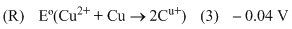

Eo (Fe3+, Fe2+) = + 0.77 V ;
Eo (Fe2+, Fe) = - 0.44 V ;
Eo (Cu2+, Cu) = + 0.34 V ;
Eo (Cu+, Cu) = + 0.52 V ;
Eo (O2(g) + 4H+ + 4e- → 2H2O] = +1.23 V ;
Eo [(O2(g) + 2H2O + 4e- → 4OH- )] = +0.40 V ;
Eo (Cr3+, Cr) = -0.74 V ;
Eo (Cr2+, Cr) = -0.91 V ;
Match Eo of the redox pair in List-I with the values given in List-II and select the correct answer using the code given below the lists :
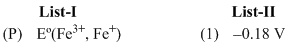
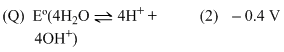
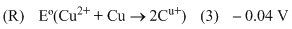

Options
A.P → 4, Q → 1, R → 2, S → 3
B.P → 2, Q → 3, R → 4, S → 1
C.P → 1, Q → 2, R → 3, S → 4
D.P → 3, Q → 4, R → 1, S → 2
Solution
Detailed solution available for registered users.
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
A sample of gaseous mixture in the beaker contains 6.08 × 1020 molecules of N2, 0.76 × 1020 molecules of O2, a...A substance AxBy crystallizes in a face-centred cubic (fcc) Lattice in which atoms ′A′ occupy each corner of...The Ca2+ and F- are located in Caf crystal, respectively at face centred cubic lattice points and in...For the reaction at 300 K at 1 bar2A(g) + B (g) → 2C(g)ᐃHof [A(g)] = -110 calᐃHof [B(g)] = - 50 cal...Given van der Waals constant for NH3, H2 and CO2 are respectively 4.17, 0.244, 1.36 and 3.59, which one of the following...