Gaseous StateHard
Question
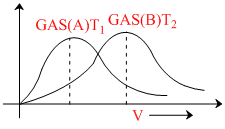
For the above graph, drawn for two different samples of gases at two different temperatures. T1 and T2, which of the following statements is/are necessarily true.
Options
A.If T2 > T1, MB is necessarily greater than MA
B.If T1 > T2, MA is necessarily greater than MB
C.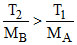
D.Nothing can be predicted
Solution
Clearly from the diagram (vMPS)B > (vMPS)A ⇒ 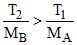 ⇒
⇒ 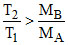
hence if T1 > T2. MA is necessarily greater than MB
hence if T1 > T2. MA is necessarily greater than MB
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
The coordination number in hcp is...In a certain sample of gas at 25oC the number of molecules having speeds between 4 km sec-1 and 4.1 km sec-1 is N. If th...The order of ionisation potential between He+ ion and H-atom (both species are in gaseous state) is :-...A sample of gaseous mixture in the beaker contains 6.08 × 1020 molecules of N2, 0.76 × 1020 molecules of O2, a...If a gas is allowed to expand at constant tempeature then which of the following does not hold true :...