Gaseous StateHard
Question
The rate of diffusion of 2 gases ′A′ and ′B′ are in the ratio 16: 3. If the ratio of their masses present in the mixture is 2 : 3. Then
Options
A.The ratio of their molar masses is 16 : 1
B.The ratio of their molar masses is 1 : 4
C.The ratio of their moles present inside the container is 1 : 24
D.The ratio of their moles present inside the container is 8 : 3
Solution
Given 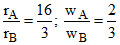
we have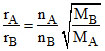
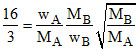
=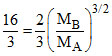 ⇒
⇒ 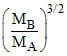 = 8
= 8
⇒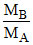 = 4
= 4
∴ mole ratio =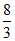
we have
=
⇒
∴ mole ratio =
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
What is the cordination number of sodium in Na2O ?...The mass of molecule A is twice that of molecule B. The root mean square velocity of molecule A is twice that of molecul...If a gas is expanded at constant temperature :...The coordination number of a metal crystallizing in a hexagonal close packed structure is :...Which one of the following statements is NOT true about the effect of an increase in temperature on the distribution of ...