Gaseous StateHard
Question
If a mixture containing 3 moles of hydrogen and 1 mole of nitrogen is converted completely into ammonia, the ratio of initial and final volume under the same temperature and pressure would be :
Options
A.3 : 1
B.1 : 3
C.2 : 1
D.1 : 2
Solution
Initial N2 + 3H2 → 2NH3
1 3
final - - 2
ratio =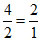
1 3
final - - 2
ratio =
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
The compressibility factor for a real gas at highpressure is :-...SO2 gas is used as a bleaching agent it′s bleaching action is :-...Equal weight of SO2 and O2 are mixed in an container at 25oC. The fraction of the total pressure exerted by O2 is :-...A hypothetical gaseous element having molecular formula Mx may be changed to another gaseous allotrope having molecular ...The compressibility factor for an ideal gas is ?...