SHMHard
Question
The initial rate of hydrolysis of methyl acetate (1M) by a weak acid (HA, 1M) is 1/100th of that of a strong acid (HX, 1M), at 25oC. The Ka of HA is
Options
A.
1 10-4
B.
1 10-5
C.
1 10-6
D.
1 10-3
Solution
Rate law for acid catalysed hydrolysis of ester is
rate = k [H+] [ester]
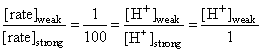
[H+]weak =0.01
HA ⇋ H+ + A-
C-x x x
Create a free account to view solution
View Solution FreeMore SHM Questions
The total energy of a harmonic oscillator of mass 2kg is 9 joules. If its energy at rest is 5 joules, its K.E. at the me...Two pendulums at rest start swinging together. Their lengths are respectively 1.44 m and 1 m. They will again start swin...A particle executes SHM on a line 8 cm long. Its K.E. and P.E. will be equal when its distance from the mean position is...A mass m is suspended from a spring of length l and force constant K. The frequency of vibration of mass is f1. The spri...The acceleration due to gravity at height R above the surface of the earth is The periodic time of a simple pendulum in ...