SHMHard
Question
The hyperconjugative stbilities of tert-butyl cation and 2-butene respectively, are due to
Options
A.
σ → P (empty) and σ → π* electron delocalisations
B.σ → σ* and σ → π electron delocalisations
C.σ → p (filled) and σ → π electron delocalisations
D.
p (filled) → σ* and σ → π* electron delocalisations
Solution
Hyperconjugative stabilities of ter-butyl cation [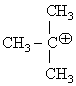 ] and 2-Butene [CH3-CH=CH-CH3]
] and 2-Butene [CH3-CH=CH-CH3]
involves 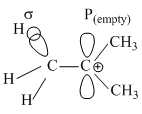 and
and 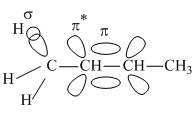 respectively
respectively
π*=Antibonding molecular orbital
π=Bonding molecular orbital
Create a free account to view solution
View Solution FreeMore SHM Questions
Two masses m1 and m2 are suspended together by a massless spring of spring constant k. When the masses are in equilibriu...In a spring-mass system, the length of the spring is L and spring constant k and it has a mass M attached to it and osci...Equation of SHM is x = 10 sin 10πt. Find the distance between the two points where speed is 50π cm/sec. x is i...If the distance between the centre of gravity and point of suspension of a compound pendulum is l and the radius of gyra...A clock S works on the oscillations of a spring. Another clock P works on the oscillations of pendulum both the clocks k...