Gaseous StateHard
Question
A quantity of gas is collected in a graduated tube over the mercury. The volume of gas at 18oC is 50 ml and the level of mercury in the tube is 100 mm above the outside mercury level. The barometer reads 750 torr. Hence , volume at S.T.P. is approximately :
Options
A.22 ml
B.40 ml
C.20 ml
D.44 ml
Solution
Net pressure of gas = Pgas
Pgas = 650 mm.
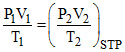
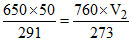
V2 = 40.11 ml P1 = 9 atm P2 = 6 atm
V1 = 5l V2 = 10 l
Pgas = 650 mm.
V2 = 40.11 ml P1 = 9 atm P2 = 6 atm
V1 = 5l V2 = 10 l
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
A teacher enters a classroom from front door while a student from back door. There are 13 equidistant rows of benches in...SO2 gas is used as a bleaching agent it′s bleaching action is :-...The compressibility factor for a real gas at highpressure is :-...The compressibility of a gas is less than unity at STP. Therefore,...Which of the following isothermal plots for fixed mass of ideal gas is / are correct ?...