Chemical Kinetics and Nuclear ChemistryHard
Question
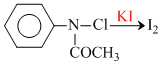
Kinetics can be studied by titration using :
Options
A.Na2S2O3
B.KMnO4
C.H2C2O4
D.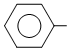 OH
OH
 OH
OHSolution
Na2S2O3 which react with I2. (Iodometric titration)
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
For the reaction A + 2B → C, rate is given by R = [A] [B]2 then the order of the reaction is...The initial rate of hydrolysis of methyl acetate (1M) by a weak acid (HA, 1M) is 1/100th of that of a strong acid (HX, 1...Which of the following statements are correct ?...In most case, for a rise of 10K temperature the rate constant is doubled to tripled. This is due to the reason that...Anisotope is :...