Coordination CompoundHard
Question
The complex showing a spin-only magnetic moment of 2.82 B.M. is
Options
A.Ni(CO)4
B.[NiCl4]2-
C.Ni(PPh3)4
D.[Ni(CN)4]2-
Solution
[NiCl4]-2, O.S. of Ni = + 2
Ni(28) = 3d8 4s2
Ni+2 = 3d8
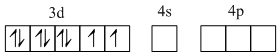
Cl- being weak ligand
No. of unpaired electrons = 2
Magnetic moment μ = 2.82 BM.
Ni(28) = 3d8 4s2
Ni+2 = 3d8
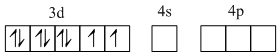
Cl- being weak ligand

No. of unpaired electrons = 2
Magnetic moment μ = 2.82 BM.
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
The complex [Cr(H2O)4Br2]Cl gives the test for :...What is the shape of Fe(CO)5 molecule ? Given that its dipole moment = 0....Which of the following pairs of structures shows geometrical isomerism ?...Among the following complexes (K-P) which compounds are diamagnetic.K3 [Fe(CN)6] (K), [Co(NH3)6] Cl3 (L), Na3 [Co(oxalat...The correct IUPAC name for the compound [Co(NH3)4CI(ONO)]Cl is :...