Chemical Kinetics and Nuclear ChemistryHard
Question
Following reaction can take place in both direction A 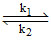 B,
B,
It is given that for the forward reaction :
and for the backward reaction
[B] Rate
0.01 M 1 × 10-2 Ms-1
0.02 M 2 × 10-2 Ms-1
Hence, net reaction rate of B is :
It is given that for the forward reaction :
and for the backward reaction
[B] Rate
0.01 M 1 × 10-2 Ms-1
0.02 M 2 × 10-2 Ms-1
Hence, net reaction rate of B is :

Options
A.= k1 [A] - k2 [B]
B.= k1 - k2 [B]
C.= k1 [A] - k2
D.= k1 - k2
Solution
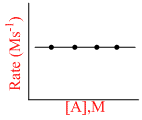
Rate of reaction independent of conc. of A
Net Rxn Rate = K1 [A]0 - K2[B]1
order wrt B is 1 because rate doubles when conc. of B doubles,
then Net Rxn Rate = k1 - k2 [B].
Net Rxn Rate = K1 [A]0 - K2[B]1
order wrt B is 1 because rate doubles when conc. of B doubles,
then Net Rxn Rate = k1 - k2 [B].
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
The integrated rate equation is Rt = log C0 - log Ct. The straight line graph is obtained by plotting...α - Particles can be detected using...Select the correct statement(s) -...The rate equation for the reaction 2A + B → C is found to be: rate k[A][B]. The correct statement in relation to t...For the elementary reaction M → N, the rate of disappearance of M increases by a factor of 8 upon doubling the con...