ElectrochemistryHard
Question
The reaction : 1/2H2(g) + AgCl(s) → H+(aq) + Cl-(aq) + Ag(s)
occurs in the galvanic cell -
occurs in the galvanic cell -
Options
A.Ag | AgCl(s) | KCl (soln) | AgNO3 (soln) | Ag
B.Pt | H2(g) | HCl (soln) | AgNO3 (soln) | Ag
C.Pt | H2(g) | HCl (soln) | AgCl(s) | Ag
D.Pt | H2(g) | KCl (soln) | AgCl(s) | Ag
Solution
(C) 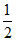 H2 → H+ + e-
H2 → H+ + e-
e- + AgCl → Ag + Cl-
_____________________
AgCl +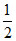 H2 → Ag + H+ + Cl-
H2 → Ag + H+ + Cl-
(D)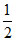 H2 → H+ + e- (KCl is neutral solution where [H+] = 10-7 M)
H2 → H+ + e- (KCl is neutral solution where [H+] = 10-7 M)
AgCl + e- Ag + Cl-
_____________________
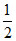 H2 + AgCl Ag + H+ + Cl-
H2 + AgCl Ag + H+ + Cl-
e- + AgCl → Ag + Cl-
_____________________
AgCl +
(D)
AgCl + e- Ag + Cl-
_____________________
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
Red hot carbon will remove oxygen from the oxide AO and BO but not from MO, while B will remove oxygen from AO. The acti...H2O2 can be prepared by successive reactions:2NH4HSO4 → H2 + (NH4)2S2O8(NH4)2S2O8 + 2H2O → 2NH4HSO4 + H2O2The first reac...These are three canonical structures of naphthalene. Examine them and find correct statement among the following :...Oxidation is :...Zn + Cu2+(aq) ⇋ Cu + Zn2+(aq)Reaction quotient is Q = . Variation of Ecell with log Q is of the type with OA = 1.1...