NEETElectrochemistryHard
Question
For the cell (at 298 K) Ag(s) | AgCl(s) | Cl- (aq) || AgNO3 (aq) | Ag(s)
Which of following is correct -
Which of following is correct -
Options
A.The cell emf will be zero when [Ag+]a = [Ag+]c ([Ag+] in anodic compartment = [Ag+] in cathodic compartment)
B.The amount of AgCl(s) precipitate in anodic compartment will decrease with the working of the cell.
C.The concentration of [Ag+] = constant, in anodic compartment during working of cell.
D.Ecell = EoAg+/Ag - EoCl-|AgCl|Ag - 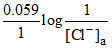
Solution
ECell = EoCell - 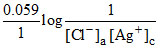 ; At [Ag+]c = [Ag+]a equilibrium will achieve and K = Ksp = [Ag+]a[Cl-]a
; At [Ag+]c = [Ag+]a equilibrium will achieve and K = Ksp = [Ag+]a[Cl-]a
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
Using the standard potential values given below, decide which of the statements I, II, III, IV are correct. Choose the r...The Gibbs energy for the decomposition of Al2O3 at 500oC is as follows: 2/3 Al2O3 → 4/3 Al + O2 ; ᐃrG = + 96...Although aluminium is above hydrogen in the electrochemical series, it is stable in air and water. Why?...A solution containing 1.0 M each of Cu(NO3)2, Mg(NO3)2, AgNO3, Hg(NO3)2 is being electrolysed using inert electrodes. Th...Standard reduction potentials of the half reactions are given below: F2(g) + 2e- → 2F-(aq) ; Eo = + 2.85 V Cl2(g) ...