ElectrochemistryHard
Question
Consider the following Galvanic cell as shown in figure.
By what value the cell voltage change when concentration of ions in anode and cathodic compartments both increased by factor of 10 at 298 K
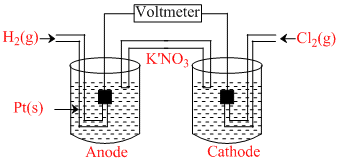
By what value the cell voltage change when concentration of ions in anode and cathodic compartments both increased by factor of 10 at 298 K

Options
A.+ 0.0591
B.- 0.0591
C.- 0.1182
D.0
Solution
Ecell = Eocell - 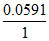 log10[H+] [Cl-] and E′cell = -
log10[H+] [Cl-] and E′cell = - 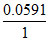 log10 100[H+] [Cl-].
log10 100[H+] [Cl-].
E′cell - Ecell = - 2 × 0.0591 = - 0.1182.
E′cell - Ecell = - 2 × 0.0591 = - 0.1182.
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
The standard EMF of a Daniel cell at 298 K is E1. When the concentration of ZnSO4 is 1.0 M and that of CuSO4 is 0.01 M, ...The equivalent conductances of two strong electrolytes at infinite dilution in H2O (where ions move freely through a sol...For the process: Cu2+ + 2e− → Cu; log[Cu2+] vs. Ered graph is shown in the figure, where OA = 0.34 V. The electrode pote...A dilute aqueous solution of Na2SO4 is electrolysed using platinum electrodes. The products at the anode and cathode are...Two weak acid solutions HA1 and HA2 each with the same concentration and having pKa values 3 and 5 are placed in contact...