ElectrochemistryHard
Question
Zn + Cu2+ (aq) ⇋ Cu + Zn2+ (aq) Reaction quotient is Q = 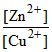 . Eocell = 1.10 V. Ecell will be 1.1591 V when :
. Eocell = 1.10 V. Ecell will be 1.1591 V when :

Options
A.[Cu2+]/[Zn2+] = 0.01
B.[Zn2+]/[Cu2+] = 0.01
C.[Zn2+]/[Cu2+] = 0.1
D.[Zn2+]/[Cu2+] = 1
More Electrochemistry Questions
ᐃG = ᐃH - TᐃS and ᐃG = ᐃH + T then is :...The highest electrical conductivity of the following aqueous solutions is of...When a lead storage battery is discharged...A depolarizer used in dry cell batteries is...When the electric current is passed through a cell having an electrolyte, the positive ions move towards cathode and neg...