Current Electricity and Electrical InstrumentHard
Question
A gaseous hydrocarbon gives upon combustion 0.72 g of water and 3.08 g of CO2.The empirical formula of the hydrocarbon is
Options
A.C2H4
B.C3H4
C.C6H5
D.C7H8
Solution
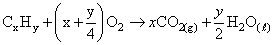
moles of
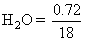 = 0.04
= 0.04moles of
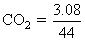 = 0.07
= 0.07∴
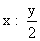 = 0.07 :0.04
= 0.07 :0.04⇒ x : y = 0.07 : 0.08 ⇒ 7 : 8
Ratio of C & H is x : y
∴ C : H = 7 : 8
Therefore empirical formula of hydrocarbon should be C7H8.
Create a free account to view solution
View Solution FreeTopic: Current Electricity and Electrical Instrument·Practice all Current Electricity and Electrical Instrument questions
More Current Electricity and Electrical Instrument Questions
Eels are able to generate current with biological cells called electroplaques . Theelectroplaques The electroplaques is ...A diode detector is used to detect an amplitude modulated wave of 60% modulation by using a condenser of capacity 250 pi...In an a. c. circuit, the instantaneous e.m.f. and current are given by e = 100 sin 30 t i = 20 sin 30t-π4 In one cy...If E = 200Volt, R = 25Ω, L = 2H and C = 2μF and the frequency is variable then the current at f = 0 and f = &#...Constanton wire is used in making standard resistances, because its :-...