Atomic StructureHard
Question
The species which has its fifth ionisation potential equal to 340 V is
Options
A.B+
B.C+
C.B
D.C
Solution
I.P. = 340 V so, I.E. = 340 eV = 13.6 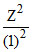
so, Z2 = 25 so, Z = 5 Therefore, (B) is correct option.
so, Z2 = 25 so, Z = 5 Therefore, (B) is correct option.
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
Deuterium nucleus contains :...Which of the following is not a fundamental particle?...In Bohr series of lines of hydrogen spectrum, the third line from the red end corresponds to which one of the following ...Bohr′s theory is not applicable to...The ratio of specific charges of α–particle and deuteron is...