Atomic StructureHard
Question
What are the values of the orbital angular momentum of an electron in the orbitals 1s, 3s, 3d and 2p -
Options
A.0, 0, 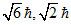
B.1, 1, 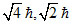
C.0, 1, 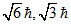
D.0, 0, 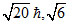
Solution
For 1s, 3s, 3d and 2p orbital, l = 0, 0, 2, 1 respectively.
Orbital angular momentum =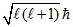 .
.
Orbital angular momentum =
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
Figure 1. electron probability density for 2 s orbitalFigure 2. wave function for 2s orbitalWhich of the following point...The binding energy of an electron in the ground state of hydrogen-like ions in whose spectrum, the third line of the Bal...Which of the following particle is not deflected in the magnetic field?...Which of the following complex ions has electrons that are symmetrically filled in both t2g and eg orbitals ?...Photons of equal energy were incident on two different gas samples. One sample containing H-atoms in the ground state an...