Atomic StructureHard
Question
If n and l are respectively the principal and azimuthal quantum numbers, then the expression for calculating the total number of electrons in any orbit is-
Options
A.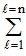 2(2l + 1)
2(2l + 1)
B.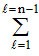 2(2l + 1)
2(2l + 1)
C.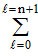 2(2l + 1)
2(2l + 1)
D.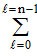 2(2l + 1)
2(2l + 1)
Solution
Total number of electrons in an orbital = 2 (2l +1).
The value of l varies from 0 to n - 1.
∴ Total numbers of electrons in any orbit =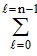 2(2l + 1).
2(2l + 1).
The value of l varies from 0 to n - 1.
∴ Total numbers of electrons in any orbit =
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
Deuterium nucleus contains :...Incorrect statement is :-...What are the number of atoms per unit cell and the number of nearest neighbours in a body centered cubic structure?...The species which has its fifth ionisation potential equal to 340 V is...Quantum numbers of an atom can be defined on the basis of...