Atomic StructureHard
Question
The difference between the wave number of 1st line of Balmer series and last line of paschen series for Li2+ ion is :
Options
A.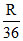
B.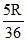
C.4R
D.R/4
Solution
For 1st line of Balmer series
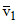 = RH(3)2
= RH(3)2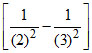 = 9R
= 9R 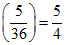 R
R
For last line of Pachen series
For last line of Pachen series
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
The triad of nuclei that is isotonic is...What is the maximum number of orbitals that can be identified with the following quantum numbers? n = 3, l = l, ml = 0...If two py - orbital from two atoms are forming π - bond, then predict their approach axis-...The quantum number ′m′ of a free gaseous atom is associated with :...The configuration 42H > 73Ne > 94 Be shows :...