Atomic StructureHard
Question
A photon of 300 nm is absorbed by a gas and then emits two photons. One photon has a wavelength 496 nm then the wavelength of second photon in nm :
Options
A.759
B.859
C.959
D.659
Solution
Eabsorbed = Eemitted
∴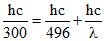
∴ λ = = 759 nm.
∴
∴ λ = = 759 nm.
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
For the same electronic transition in the following atom or ion, the frequency of the emitted radiation will be maximum ...The following electronic transitions occur when Lithium atoms are sprayed into a hot flame;Which of these transition wou...In which reaction hybridization of underlined atom does not changed :-...The wave number of electromagnetic radiation emitted during the transition of electron in between two levels of Li2+ ion...Electrons are de-exciting from the fifth orbit in hydrogen atoms but the first orbit is not available for them. The maxi...