Atomic StructureHard
Question
Correct order of radius of the Ist orbit of H, He+, Li2+, Be3+ is :
Options
A.H > He+ > Li2+ > Be3+
B.Be3+ > Li2+ > He+ > H
C.He+ > Be3+ > Li2+ > H
D.He+ > H > Li2+ > Be3+
Solution
rα 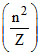 As Z increases, radius of I orbit decreases.
As Z increases, radius of I orbit decreases.
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
Which of the following graph represents the radial probability function of 3d electron?...The radii of two of the first four Bohr′s orbits of the hydrogen atom are in the ratio 1 : 4 The energy difference...Which of the following is the most correct electron displacement for a nucleophilic reaction to take place?...The principal quantum number of an atom is related to the :...What is the most probable distance of a 1s electron in a He+ ion. The wave function for 1s orbital is given by $\Psi = \...