Current Electricity and Electrical InstrumentHard
Question
Which of the following is the wrong statement?
Options
A.ONCl and ONO- are not isoelectronic
B.O3 molecule is bent
C.Ozone is violet-black in solid state
D.Ozone is diamagnetic gas
Solution
(i) Some times, two species having samevalence shell electrons are considered asisolectronic species. Accordingly ONCl and ONO- are isoelectronic to each other.
[Otherwise all four statements are correct and no answer is available in the given options].
(ii)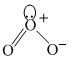 sp2, bent shaped
sp2, bent shaped
(iii)
O3(g) = Blue
O3(l) = Dark blue
O3 (s) = violet - black
(iv) All electrons are paired in structure of O3.Hence it is diamagnetic.
[Otherwise all four statements are correct and no answer is available in the given options].
(ii)
 sp2, bent shaped
sp2, bent shaped(iii)
O3(g) = Blue
O3(l) = Dark blue
O3 (s) = violet - black
(iv) All electrons are paired in structure of O3.Hence it is diamagnetic.
Create a free account to view solution
View Solution FreeTopic: Current Electricity and Electrical Instrument·Practice all Current Electricity and Electrical Instrument questions
More Current Electricity and Electrical Instrument Questions
A whetstone′s bridge is balanced with a resistance of 625 Ω in the third arm, where P, Q and S are in the 1st...Electromotive force of a cell is basically a...A piece of copper and another of germanium are colled from room temperature to 80 K. The resistance of...A galvanometer of resistance 100Ω gives full deffection for a current 10−5 A. The value of shunt required to ...What is the equivalent resistance across A and B in the figure shown, if R = 3Ω?...