Solid StateHard
Question
Given an alloy of Cu, Ag and Au in which Cu atoms constitute the CCP arrangement.If the hypothetical formula of the alloy is Cu4Ag3Au. What are the probable locations of Ag and Au atoms.
Options
A.Ag - all Tetrahedral voids; Au - all Octahedral voids
B.Ag - 3/8th Tetrahedral voids; Au - 1/4th Octahedral voids
C.Ag - 1/2 Octahedral voids; Au - 1/2 Tetrahedral voids
D.Ag - 1/2 Octahedral voids; Au - 1/2 Tetrahedral voids
Solution
Cu4 → Froms c.c.p. z = 4
Ag3 →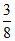 th of tetrahedral voids, [∴ No. of T- voids = 8].
th of tetrahedral voids, [∴ No. of T- voids = 8].
Au →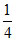 of Octahedral voids [∴ No. of O - voids = 4]
of Octahedral voids [∴ No. of O - voids = 4]
Ag3 →
Au →
Create a free account to view solution
View Solution FreeMore Solid State Questions
A solid element (monoatomic) exists in cubic crystal. If its atomic radius is 1.0 Å and the ratio of packing fraction an...Correct statement for ccp is :...The correct statement(s) regarding defects in solids is(are)...The theoretical density of ZnS is d g/cm3. If the crystal has 4% Frenkel defect, then the actual density of ZnS should b...In Na2O structure,...