Solid StateHard
Question
The ionic radii of Rb+ and I- are 1.46 and 2.16 (Ao). The most probable type of structure exhibited by it is
Options
A.CsCI type
B.NaCl type
C.ZnS type
D.CaF2 type
Solution
Radius Ratio 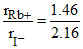 = 0.68
= 0.68
⇒ between 0.414 & 0.732
Hence Rock salt (NaCl) structure.
⇒ between 0.414 & 0.732
Hence Rock salt (NaCl) structure.
Create a free account to view solution
View Solution FreeMore Solid State Questions
A solid has a structure in which W atoms are located at the corners of a cubic lattice. O atom at the center of the edge...Number of 3rd nearest neighbour atom in a FCC unit cell are -...In a face centerd lattice of X and Y, X atoms are present at the corners while Y atoms are at face centers. Then the for...A binary solid (A+ B−) has a rock salt structure. If the edge length is 400 pm and the radius of cation is 75 pm, then t...You are given 6 identical balls.What is the maximum number of square voids and triangular voids(in separate arrangements...