Solid StateHard
Question
A solid has a structure in which W atoms are located at the corners of a cubic lattice. O atom at the center of the edges and Na atom at center of the cube. The formula for the compound is
Options
A.NaWO2
B.NaWO3
C.Na2WO3
D.NaWO4
Solution
Na = 1 × 1 = 1
W = 8 ×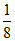 = 1 ⇒ NaWO3
= 1 ⇒ NaWO3
O = 12 ×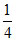 = 3.
= 3.
W = 8 ×
O = 12 ×
Create a free account to view solution
View Solution FreeMore Solid State Questions
Which of the following substances acts as a superconductor at approximately 4 K?...If the anions (A) form hexagonal closest packing and cations (C) occupy only 2/3 octahedral voids in it, then the genera...TiO2 is a well-known example of...Which of the following statements is/are false....The number of octahedral voids per unit BCC cell is...