Nuclear Physics and RadioactivityHard
Question
de-Broglie wavelength of an electron in the nth Boher orbit is λn and the angular momentum is Jn, then.
Options
A.Jn ∝ λn
B.λn ∝ 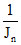
C.λn ∝ Jn2
D.None of these
Solution
Jn = 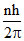 ⇒ nλν = 2π 2(r0n2)
⇒ nλν = 2π 2(r0n2)
So Jn ∝ λn ⇒ λn = (2πr0)n
So Jn ∝ λn ⇒ λn = (2πr0)n
Create a free account to view solution
View Solution FreeMore Nuclear Physics and Radioactivity Questions
A nucleus undergoes a series of decay according to the scheme A B C D E Atomic number and mass numbers of E are 69 and 1...When the atomic number Z of the nucleus increase...The β-particles are emmitted by :-...Light of two different frequencies whose photons have energies of 1eV and 2.5 eV are incident one by one on a metal surf...Choose the correct statement(s) for hydrogen and deuterium atoms (considering the motion of nucleus)...