Atomic StructureHard
Question
The wavelength of visible light is:
Options
A.2000 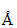 - 3700
- 3700 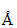
B.7800 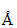 - 8900
- 8900 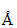
C.3800 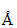 - 7600
- 7600 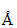
D.None of these
Solution
Wavelength of visible light is 3800 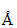 - 7600
- 7600 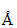
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
For a d-electron, the orbital angular momentum is :...The fraction of volume occupied by the nucleus with respect to the total volume of an atom is...In order to increase the kinetic energy of ejected photoelectrons, there should be an increase in...The electron in the same orbital may be identified with the quantum number...Which of the following cannot be circumference of an orbit in H-atom? (r0 = radius of the first orbit)...