Solid StateHard
Question
Platinum crystallizes in a face-centered cubic crystal with a unit cell length ′a′. The distance between nearest neighbors is :
Options
A.a
B.a 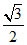
C.a 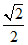
D.a 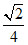
Solution
Distance between nearest neighbours is along the face diagonal = 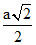
Create a free account to view solution
View Solution FreeMore Solid State Questions
In a solid lattice, the cation has left a lattice site and is located at interstitial position, the lattice defect is :...Zinc Oxide, white in colour at room temperature, acquires yellow colour on heating due to:...In a face centred cubic lattice, atom A occupies the corner positions and atom B occupies the face centre positions. If ...Lithium has a bcc structure. Its density is 530 kg m-3 and its atomic mass is 6.94 g mol-1. Calculated the edge length o...Consider a cube 1 of Body Centered Cubic unit cell of edge length a now atom at the body center can be viewed to be lyin...