SolutionHard
Question
On mixing, heptane and octane form an ideal solution. At 373 K, the vapour pressures of the two liquid components (heptane and octane) are 105 kPa and 45 kPa respectively. Vapour pressure of the solution obtained by mixing 25.0 g of heptane and 35 g of octane will be (molar mass of heptane= 100 g mol-1 and of octane = 114 g mol-1).
Options
A.72.0 kPa
B.36.1 kPa
C.96.2 kPa
D.144.5 kPa
Solution
Mole fraction of Heptane = 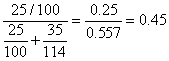
XHeptane = 0.45
∴ Mole fraction of octane = 0.55 = Xoctane
Total pressure =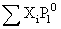
= (105 × 0.45) + (45 × 0.55) kPa
= 72.0 KPa
Mole fraction of Heptane =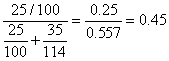
XHeptane = 0.45
∴ Mole fraction of octane = 0.55 = Xoctane
Total pressure =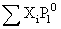
= (105 × 0.45) + (45 × 0.55) kPa
= 72.0 KPa
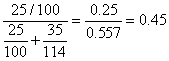
XHeptane = 0.45
∴ Mole fraction of octane = 0.55 = Xoctane
Total pressure =
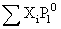
= (105 × 0.45) + (45 × 0.55) kPa
= 72.0 KPa
Mole fraction of Heptane =
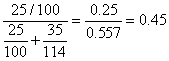
XHeptane = 0.45
∴ Mole fraction of octane = 0.55 = Xoctane
Total pressure =
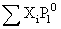
= (105 × 0.45) + (45 × 0.55) kPa
= 72.0 KPa
Create a free account to view solution
View Solution FreeMore Solution Questions
For each of the following dilute solutions, van’t Hoff factor is equal of 3, except...Density of 1M solution of a non-electrolyte C6H12O6 is 1.18 g/mL. If Kf (H2O) is 1.86o mol-1 kg, solution freezes at :...The osmotic pressures of 0.010 M solutions of KI and of sucrose (C12H22O11) are 0.432 atm and 0.24 atm respectively. The...Which of the following azeotropic solutions has the b.p. less than b.p. of the constituents A and B ?...The normal freezing point of nitrobenzene is 278.82 K. A non-volatile solute is dissolved in it and a solution of molali...