Nuclear Physics and RadioactivityHard
Question
Choose the correct statement(s) for hydrogen and deuterium atoms (considering the motion of nucleus)
Options
A.The radius of first Bohr orbit of deuterium is less than that of hydrogen
B.The speed of electron in first Balmer line of deuterium is more than that of hydrogen
C.The wave length of first Balmer line of deuterium is more than that of hydrogen
D.The angular momentum of electrom in the first Bohr orbit of deuterium is more than that of hydrogen
Solution
We have r ∝ 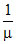 , μ = reduced massWe have r ∝
, μ = reduced massWe have r ∝ 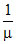 , μ = reduced mass
, μ = reduced mass
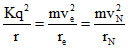 ; mvere =
; mvere = 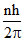
Create a free account to view solution
View Solution FreeMore Nuclear Physics and Radioactivity Questions
A radioactive sample disintegrates by 10% during one month. How much fraction will disintegrate in four months :-...A radioactive decay equation is giventhen which is incorrect :-...If 92U238 → ZXA + 2He4 then Z and A will be :...If radius of nucleus is estimated to be 3.6 Fermi then the radius nucleus be nearly...A radioactive reaction is 92U238 → 82Pb206. How many α and β particles are emitted ?...